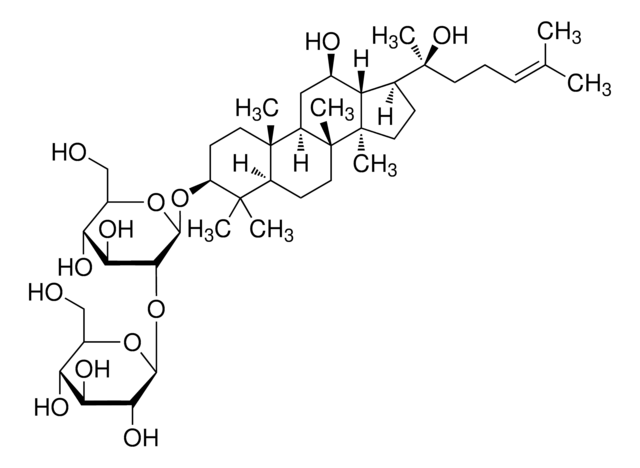
SMB00348
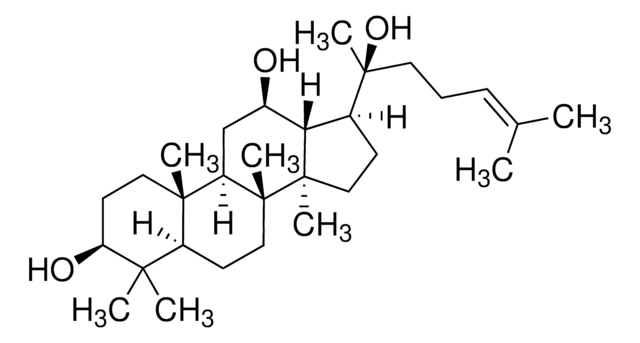
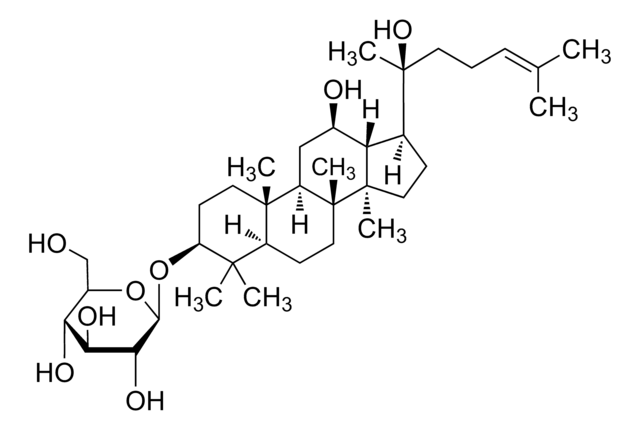
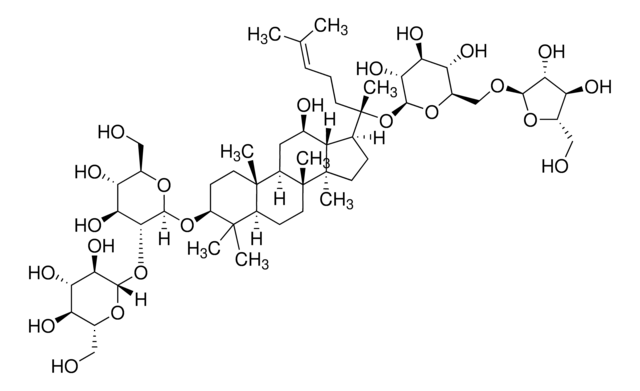
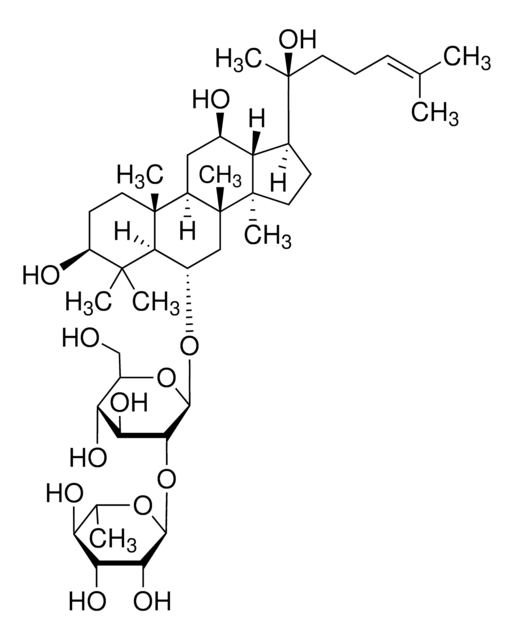
Ginsenosid K
≥96% (HPLC)
Synonym(e):
Protopanaxadiol-20-O-glucosid
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
≥96% (HPLC)
Form
powder
Anwendung(en)
metabolomics
vitamins, nutraceuticals, and natural products
SMILES String
CC(C)=CCC[C@](C1C2[C@H](O)CC3[C@@]([C@]2(C)CC1)(C)CCC4C(C)(C)[C@@H](O)CC[C@@]43C)(C)O[C@@H]5O[C@H](CO)[C@@H](O)[C@H](O)[C@H]5O
InChI
1S/C36H62O8/c1-20(2)10-9-14-36(8,44-31-30(42)29(41)28(40)23(19-37)43-31)21-11-16-35(7)27(21)22(38)18-25-33(5)15-13-26(39)32(3,4)24(33)12-17-34(25,35)6/h10,21-31,37-42H,9,11-19H2,1-8H3/t21?,22-,23-,24?,25?,26+,27?,28-,29+,30-,31+,33+,34-,35-,36+/m1/s1
InChIKey
FVIZARNDLVOMSU-KJULZEBLSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Verwandte Kategorien
Allgemeine Beschreibung
Anwendung
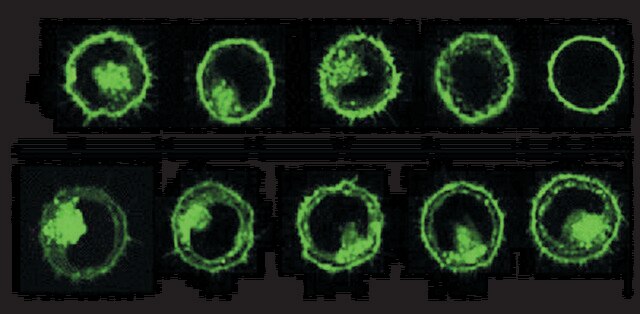
- Combination of Electrochemistry and Mass Spectrometry to Study Nitric Oxide Metabolism and Its Modulation by Compound K in Breast Cancer Cells.: This research explores the interaction of Ginsenoside Compound K with nitric oxide metabolism in breast cancer cells, employing a combination of electrochemical techniques and mass spectrometry to elucidate the metabolic pathways and potential therapeutic effects of Compound K (Zhao et al., 2022).
- Metabolic analysis of Panax notoginseng saponins with gut microbiota-mediated biotransformation by HPLC-DAD-Q-TOF-MS/MS.: This article investigates the gut microbiota-mediated biotransformation of Panax notoginseng saponins, including Compound K, detailing the complex interactions and metabolic profiles in a biological system, which could inform their therapeutic use and bioavailability (Chen et al., 2018).
Biochem./physiol. Wirkung
Signalwort
Warning
H-Sätze
Gefahreneinstufungen
Acute Tox. 4 Oral
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.