17793
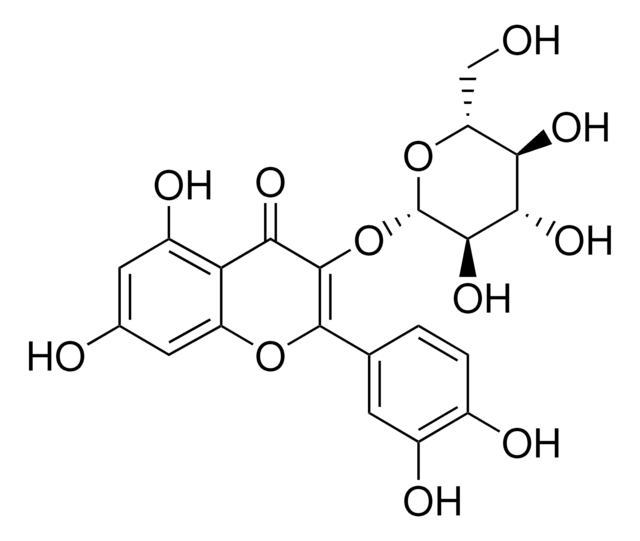
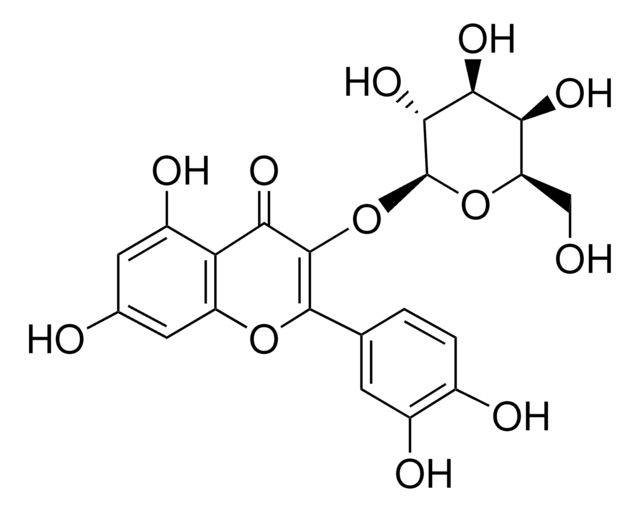
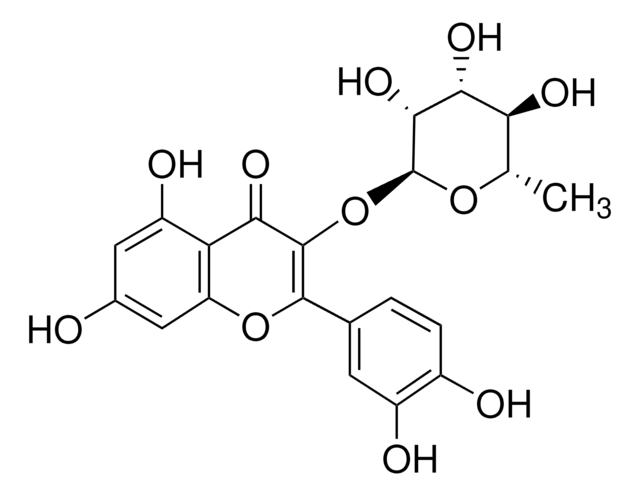
Quercetin-3-β-D-glucosid
≥90% (HPLC)
Synonym(e):
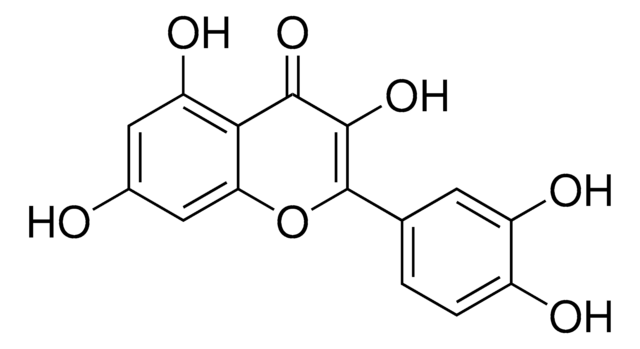
3,3′,4′,5,7-Pentahydroxy-flavon-3-β-glucosid, Isoquercitrin
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
≥90% (HPLC)
Anwendung(en)
metabolomics
vitamins, nutraceuticals, and natural products
Lagertemp.
−20°C
SMILES String
OC[C@H]1O[C@@H](OC2=C(Oc3cc(O)cc(O)c3C2=O)c4ccc(O)c(O)c4)[C@H](O)[C@@H](O)[C@@H]1O
InChI
1S/C21H20O12/c22-6-13-15(27)17(29)18(30)21(32-13)33-20-16(28)14-11(26)4-8(23)5-12(14)31-19(20)7-1-2-9(24)10(25)3-7/h1-5,13,15,17-18,21-27,29-30H,6H2/t13-,15-,17+,18-,21+/m1/s1
InChIKey
OVSQVDMCBVZWGM-QSOFNFLRSA-N
Angaben zum Gen
mouse ... Hexa(15211)
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Verwandte Kategorien
Allgemeine Beschreibung
Anwendung
- as a dietary flavonoid supplement to check its binding capacity with human small ubiquitin-related modifier 1 (SUMO1) protein using surface plasmon resonance (SPR)
- as an inhibitor for Escherichia coli adenosine triphosphate (ATP) synthase
- as an anti-aggregation agent to test its activity against β-amyloid, green fluorescent protein (GFP), and chymotrypsinogen proteins
Biochem./physiol. Wirkung
Verpackung
Lagerklassenschlüssel
11 - Combustible Solids
WGK
WGK 3
Persönliche Schutzausrüstung
Eyeshields, Gloves, type N95 (US)
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Protokolle
HPLC Analysis of Polyphenols in Nero d'Avola Red Wine on Discovery® HS C18 (UV 280 nm)
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.