M55909
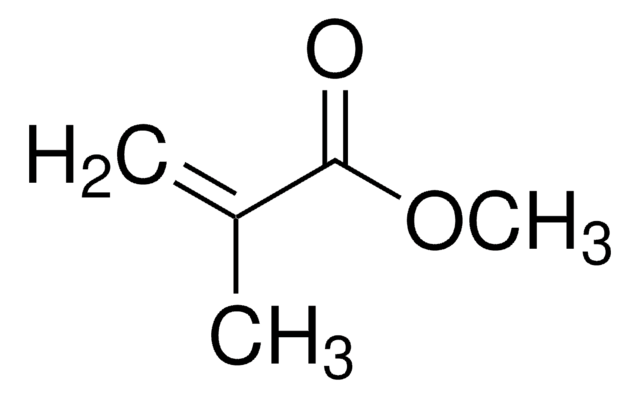
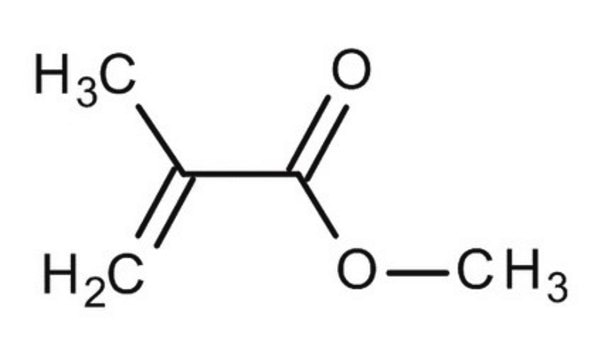
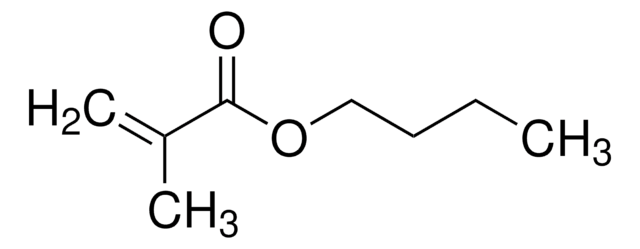
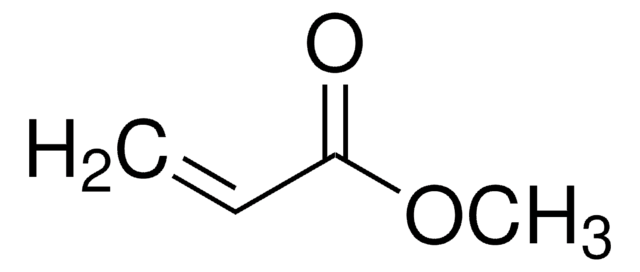
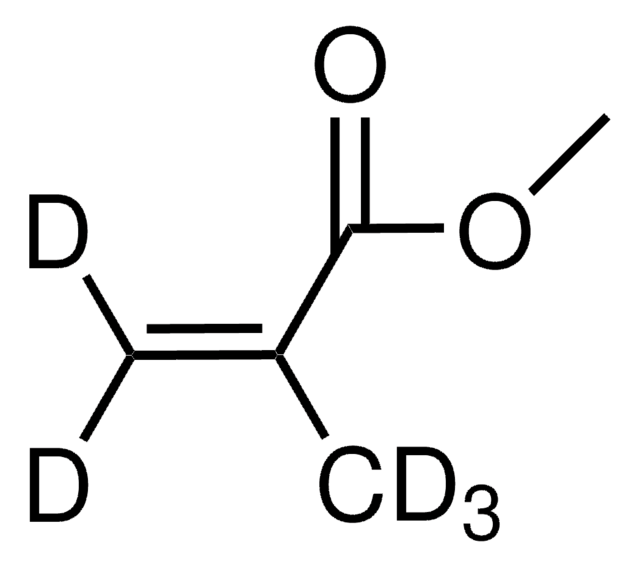
Methyl methacrylate
contains ≤30 ppm MEHQ as inhibitor, 99%
Synonym(s):
Methacrylic acid methyl ester, Methyl 2-methylprop-2-enoate, Methyl 2-methylpropenoate
About This Item
Recommended Products
vapor density
3.5 (vs air)
Quality Level
vapor pressure
29 mmHg ( 20 °C)
assay
99%
autoignition temp.
815 °F
contains
≤30 ppm MEHQ as inhibitor
expl. lim.
12.5 %
refractive index
n20/D 1.414 (lit.)
bp
100 °C (lit.)
mp
−48 °C (lit.)
density
0.936 g/mL at 25 °C (lit.)
storage temp.
2-8°C
SMILES string
COC(=O)C(C)=C
InChI
1S/C5H8O2/c1-4(2)5(6)7-3/h1H2,2-3H3
InChI key
VVQNEPGJFQJSBK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
- Lanthanide-complex grafted poly(methyl methacrylate-co-maleic anhydride) copolymer. These luminescent polymers exhibit high thermal stability and can be used as luminous layers for optoelectronic devices.
- Poly (methyl methacrylate) (PMMA), is a common material used in the production of lenses for concentrating photovoltaic (CPV) modules.
- Polymethyl methacrylate, methyl methacrylate crosspolymer, and methyl methacrylate/glycol dimethacrylate crosspolymers. These polymers are used in cosmetic surgery, dentistry, and joint replacement.
- Poly (methyl methacrylate) (PMMA)-based personalized medical devices.
- Interpenetrating methyl methacrylate-based polymeric networks with enhanced thermal and mechanical properties.
- Poly(methyl methacrylate-co-hydroxyethyl methacrylate) (PMMA-co-PHEMA) copolymers by emulsion copolymerization. These copolymers form thermooxidatively stable and ductile films.
- Poly(methyl methacrylate) nanoparticles through differential microemulsion polymerization.
signalword
Danger
Hazard Classifications
Flam. Liq. 2 - Skin Irrit. 2 - Skin Sens. 1B - STOT SE 3
target_organs
Respiratory system
Storage Class
3 - Flammable liquids
wgk_germany
WGK 1
flash_point_f
50.0 °F - closed cup
flash_point_c
10 °C - closed cup
ppe
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
RAFT (Reversible Addition Fragmentation chain Transfer) polymerization is a reversible deactivation radical polymerization (RDRP) and one of the more versatile methods for providing living characteristics to radical polymerization.
The manufacture of monomers for use in ophthalmic applications is driven by the need for higher purity, improved reliability of manufacturing supply, but ultimately by the need for the increased comfort, convenience, and safety of contact lens wearers. Daily wear contact lenses have the potential to fill this need for many customers; however, their widespread use is constrained by higher costs compared to weekly- or monthly-based lenses. New approaches that improve cost structure and result in higher quality raw materials are needed to help make contact lenses more affordable and accelerate growth of the contact lens market.
Protocols
Monodisperse, surfactant-free polymer spheres for use as colloidal crystal templates can be easily obtained in reasonably large quantities. Typical synthesis methods for poly(methyl methacrylate) (PMMA) and poly(styrene) (PS) by emulsifier free emulsion polymerization are described below and yield spheres several hundred nanometers in diameter.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service