732117
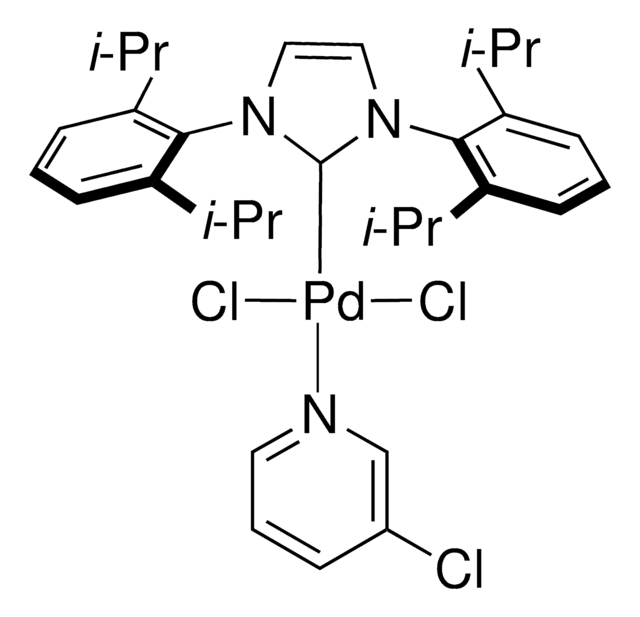
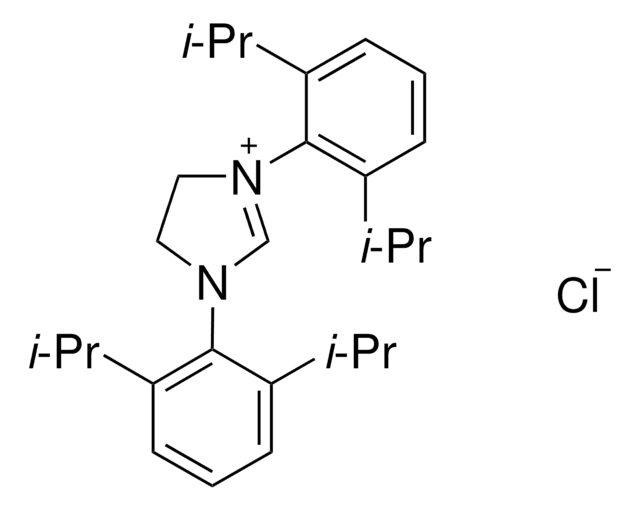
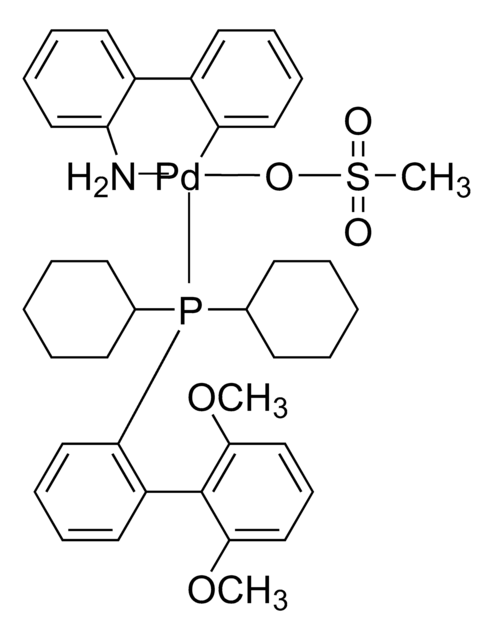
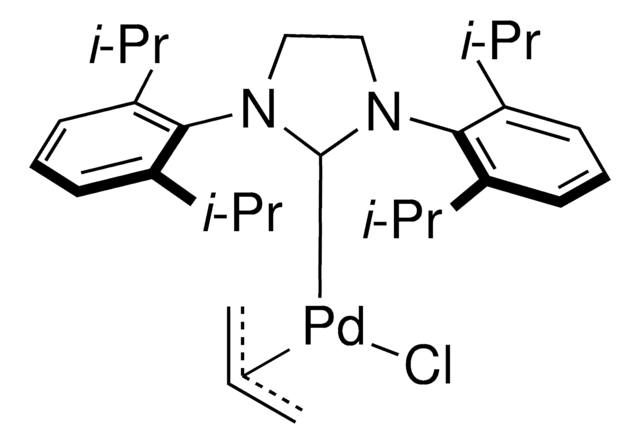
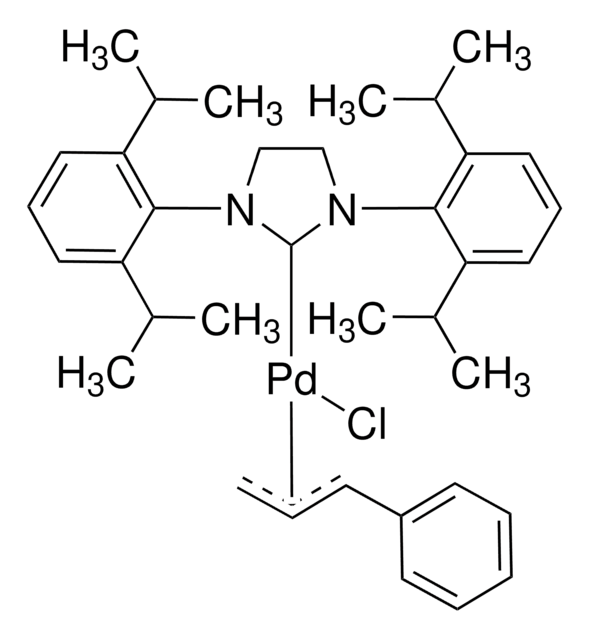
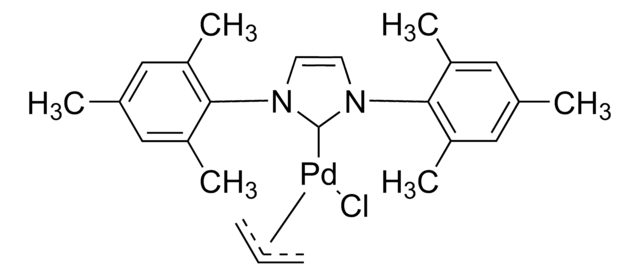
Pd-PEPPSI™-IPent catalyst
≥95%
Synonym(s):
Dichloro[1,3-bis(2,6-Di-3-pentylphenyl)imidazol-2-ylidene](3-chloropyridyl)palladium(II), [1,3-Bis(2,6-Di-3-pentylphenyl)imidazol-2-ylidene](3-chloropyridyl)dichloropalladium(II), [1,3-Bis(2,6-Di-3-pentylphenyl)imidazol-2-ylidene](3-chloropyridyl)palladium(II) dichloride
About This Item
Recommended Products
Quality Level
assay
≥95%
form
solid
reaction suitability
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Cross Couplings
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
mp
195-201 °C
storage temp.
−20°C
SMILES string
Clc1cccnc1.CCC(CC)c2cccc(C(CC)CC)c2N3C=CN(c4c(cccc4C(CC)CC)C(CC)CC)\C3=[Pd](/Cl)Cl
InChI
1S/C35H52N2.C5H4ClN.2ClH.Pd/c1-9-26(10-2)30-19-17-20-31(27(11-3)12-4)34(30)36-23-24-37(25-36)35-32(28(13-5)14-6)21-18-22-33(35)29(15-7)16-8;6-5-2-1-3-7-4-5;;;/h17-24,26-29H,9-16H2,1-8H3;1-4H;2*1H;/q;;;;+2/p-2
InChI key
BCXSKTXOKALLAZ-UHFFFAOYSA-L
Related Categories
General description
Application
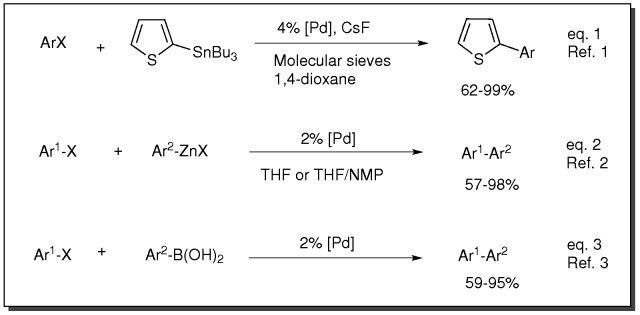
Cross-Coupling, Amination and Heck Transformation using PEPPSI Catalysts
- Catalyst for Stille coupling reaction (eq. 1)
- Catalyst for Negishi coupling reaction (eq. 2)
- Catalyst for Suzuki coupling reaction (eq. 3)
For small scale and high throughput uses, product is also available as ChemBeads (928399)
Legal Information
related product
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Professor Mike Organ and co-workers have developed the PEPPSI™ (Pyridine-Enhanced Precatalyst Preparation Stabilization and Initiation) precatalysts for palladium-catalyzed cross-coupling reactions.
Explore the higher efficiency and superior functional group tolerance of the Pyridine-Enhanced Precatalyst Preparation Stabilization and Initiation (PEPPSI™) palladium N-heterocyclic-carbene catalyst system.
All of the preformed catalysts used in the kit are air and moisture stable complexes in their commercially available form.
Multiple tools have been created to ensure your success with kit set up. Start with the more detailed guide to ensure you are comfortable with all of the steps before using the quick guides on the excel worksheet. Remember that while the technique is new, it is still organic chemistry and so the steps will seem easy once you try just one kit. It is just a new way of approaching something you are already very good at.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service