SML0562
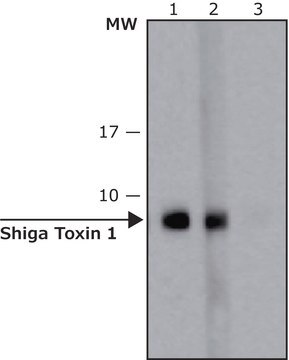
Shiga Toxin 1, B subunit
recombinant, expressed in E. coli, ≥95% (SDS-PAGE)
Synonym(s):
SLT1, STX1, STxB
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
UNSPSC Code:
12352200
NACRES:
NA.77
Recommended Products
recombinant
expressed in E. coli
Quality Level
Assay
≥95% (SDS-PAGE)
form
lyophilized
shipped in
dry ice
storage temp.
−20°C
Application
Shiga Toxin 1, B subunit has been used to induce globotriaosylceramide (Gb3) endocytosis.
Biochem/physiol Actions
The Shiga toxins are a family of related protein toxins secreted by certain types of bacteria. Shiga toxin (Stx) is produced by Shigella dysenteriae, whereas the Shiga-like toxins, Stx1 and Stx2, with a few known isoforms, are secreted by specific strains of Escherichia coli named Shiga-toxin-producing E. coli (STEC) such as E. coli O157:H7, which causes bloody diarrhea and hemorrhagic colitis in humans, sometimes resulting in fatal systemic complications.
Stx1 is identical to Stx, while the Stx2 isoforms share less sequence similarity with Stx (~60%) and are immunologically distinct. In spite of the differences in their amino acid sequence, all Stx isoforms share the same overall toxin structure and mechanism of action.
Shiga toxins consist of two polypeptides: An A chain and a B chain non-covalently associated with an apparent stoichiometry of one A and five B chains, to form the holotoxin. The catalytic A subunit has a RNA N-glycosidase activity that inhibits eukaryotic protein synthesis. The B subunits form a pentamer that recognizes and binds to the functional cell-surface receptor globotriaosylceramide [Gb3; Gala(1-4)-Galb(1-4)-Glcb1-ceramide]. Gb3 is overexpressed in membranes of numerous tumor cells, therefore STxB binding to Gb3 receptors may be useful for cell-specific vectorization, labeling and imaging purposes.
Stx1 is identical to Stx, while the Stx2 isoforms share less sequence similarity with Stx (~60%) and are immunologically distinct. In spite of the differences in their amino acid sequence, all Stx isoforms share the same overall toxin structure and mechanism of action.
Shiga toxins consist of two polypeptides: An A chain and a B chain non-covalently associated with an apparent stoichiometry of one A and five B chains, to form the holotoxin. The catalytic A subunit has a RNA N-glycosidase activity that inhibits eukaryotic protein synthesis. The B subunits form a pentamer that recognizes and binds to the functional cell-surface receptor globotriaosylceramide [Gb3; Gala(1-4)-Galb(1-4)-Glcb1-ceramide]. Gb3 is overexpressed in membranes of numerous tumor cells, therefore STxB binding to Gb3 receptors may be useful for cell-specific vectorization, labeling and imaging purposes.
Physical form
The recombinant product is Shiga toxin 1, B subunit, a 7 kDa protein containing 69 amino acid residues. It is lyophilized from 0.2 μm-filtered solution of phosphate buffer without any carrier protein.
Preparation Note
Reconstitute the contents of the vial using water to a concentration of 0.1-1.0 mg/mL. This solution can then be further diluted into other aqueous buffers and stored at 2-8 °C for up to 4 months or at −20 °C for extended use.
Analysis Note
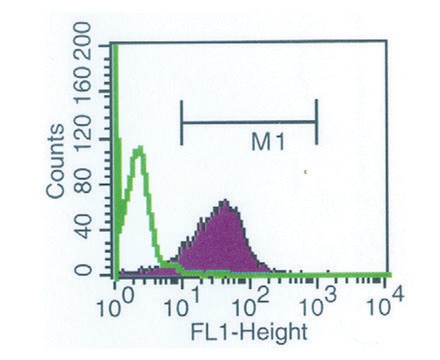
Gb3 Binding activity: significant binding above background is achieved with ≤1 μg/mL of STxB. The activity of STxB is measured by its ability to bind to Gb3, which requires its pentameric form.
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
The Pseudomonas aeruginosa lectin LecA triggers host cell signalling by glycosphingolipid-dependent phosphorylation of the adaptor protein CrkII
Zheng S, et al.
Biochimica et Biophysica Acta, 1864(7), 1236-1245 (2017)
In-Sun Shin et al.
Chemical & pharmaceutical bulletin, 54(4), 522-527 (2006-04-06)
A two-step binding assay for globotriaosylceramide (Gb3) content was developed by histidine-tagging strategy, which is a well-established method for the purification of recombinant proteins. The complete binding of the recombinant His-tagged Shiga toxin 1B subunit (1B-His) (1 microg/ml) to the
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service