E6510
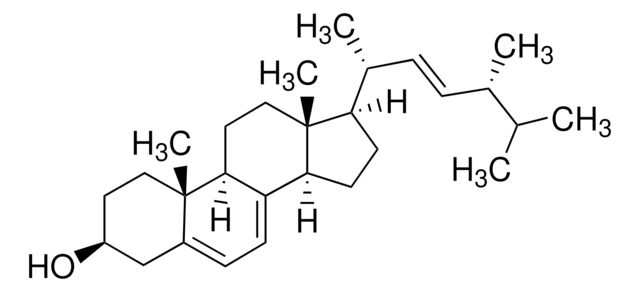
Ergosterol
≥75%
Synonym(s):
3β-Hydroxy-5,7,22-ergostatriene, 5,7,22-Ergostatrien-3β-ol, Provitamin D2
About This Item
Recommended Products
biological source
microbial
Quality Level
Assay
≥75%
form
powder
color
white to off-white
mp
156-158 °C (lit.)
storage temp.
2-8°C
SMILES string
[H][C@@]1(CC[C@@]2([H])C3=CC=C4C[C@@H](O)CC[C@]4(C)[C@@]3([H])CC[C@]12C)[C@H](C)\C=C\[C@H](C)C(C)C
InChI
1S/C28H44O/c1-18(2)19(3)7-8-20(4)24-11-12-25-23-10-9-21-17-22(29)13-15-27(21,5)26(23)14-16-28(24,25)6/h7-10,18-20,22,24-26,29H,11-17H2,1-6H3/b8-7+/t19-,20+,22-,24+,25-,26-,27-,28+/m0/s1
InChI key
DNVPQKQSNYMLRS-APGDWVJJSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- as a component of yeast cell monolayer models, to study the effects of steroidal and triterpenoid saponins on the monolayers
- as a standard sample, to isolate and identify a fungal metabolite fraction containing ergosterol
- as a component of the culture medium to isolate the CYP51RNAi phenotype of Trypanosoma brucei gambiense, to verify ergosterol biosynthesis
Biochem/physiol Actions
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 4
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Vitamin D2 (ergocalciferol) is naturally synthesized from ergosterol by invertebrates, fungi, and plants in response to ultraviolet B irradiation, while vitamin D3 synthesis (cholecalciferol) is uniquely initiated in the skin of vertebrates. During sun exposure, ultraviolet B photons are absorbed by 7-dehydrocholesterol, which is found within the plasma membranes of epidermal and dermal skin layers. This reaction yields an unstable derivative of 7-dehydrocholesterol, named precholecalcitrol, which rapidly rearranges to vitamin D3. Vitamin D binding protein (DBP) is a carrier protein responsible for drawing vitamin D3 from the plasma membrane into the dermal capillaries within the extracellular space.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service