L3900
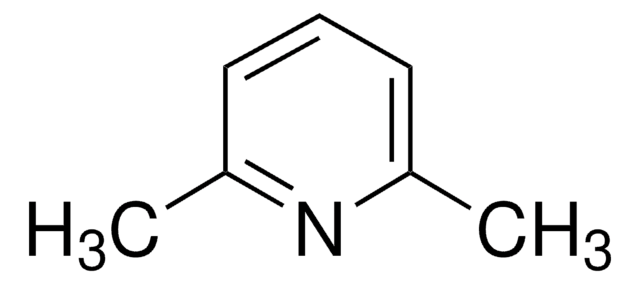
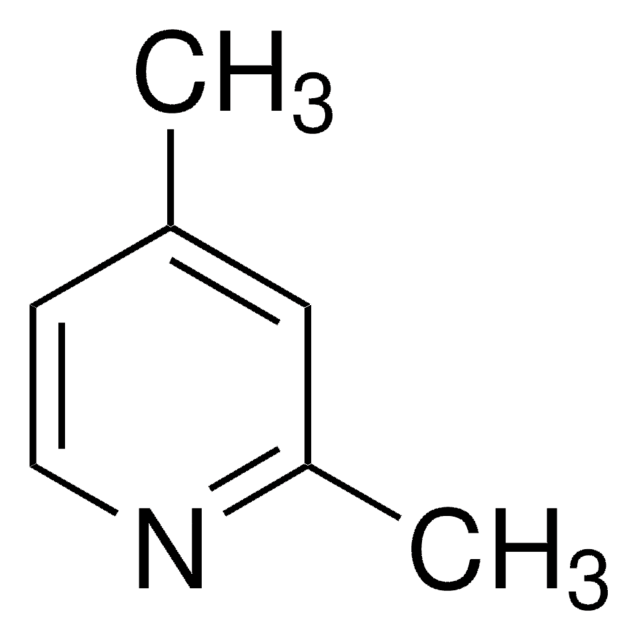
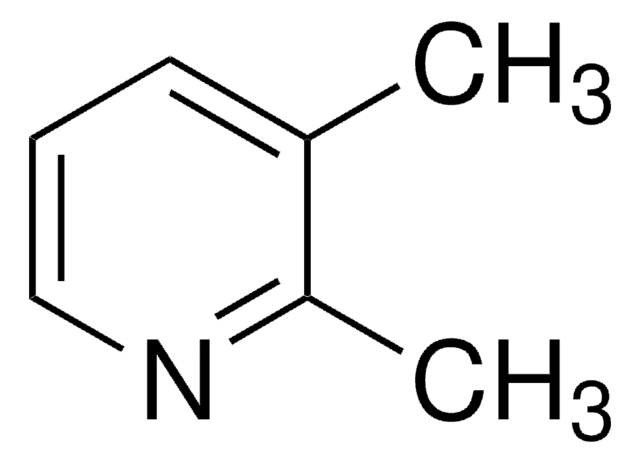
2,6-Lutidine
ReagentPlus®, 98%
Synonym(s):
2,6-Dimethylpyridine
About This Item
Recommended Products
Quality Level
product line
ReagentPlus®
Assay
98%
refractive index
n20/D 1.497 (lit.)
bp
143-145 °C (lit.)
mp
−6 °C (lit.)
density
0.92 g/mL at 25 °C (lit.)
SMILES string
Cc1cccc(C)n1
InChI
1S/C7H9N/c1-6-4-3-5-7(2)8-6/h3-5H,1-2H3
InChI key
OISVCGZHLKNMSJ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
It can be used:
- As a promoter for catalytic asymmetric fluorination of α-cyanophosphonates in the presence of chiral Pd(II)-bisphosphine complexes.
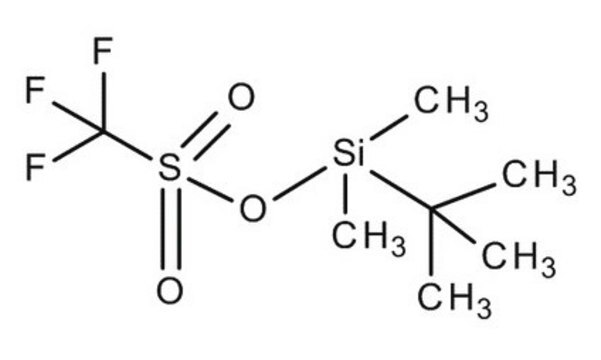
- In combination with tert-butyldimethylsilyl triflate for the protection of tertiary alcohols and unreactive secondary alcohols.
- In combination with triethylsilyl trifluoromethanesulfonate for the conversion of acetals to the corresponding aldehydes in dichloromethane followed by workup in water.
Legal Information
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Eye Irrit. 2 - Flam. Liq. 3 - Skin Irrit. 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
91.4 °F
Flash Point(C)
33 °C
Personal Protective Equipment
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
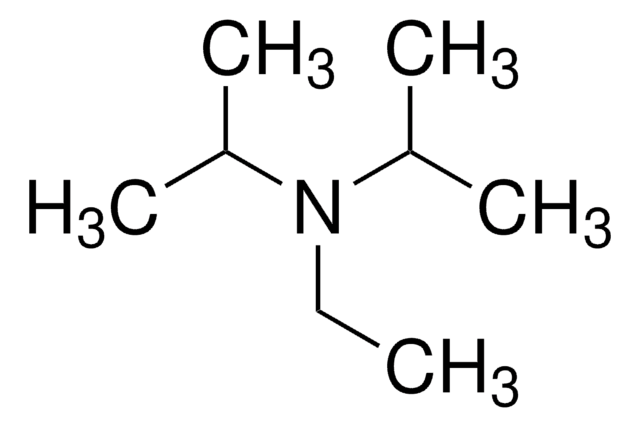
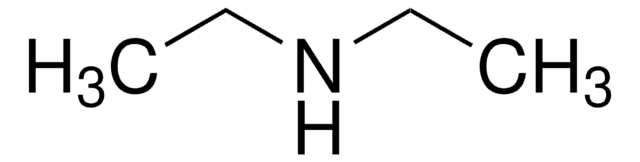
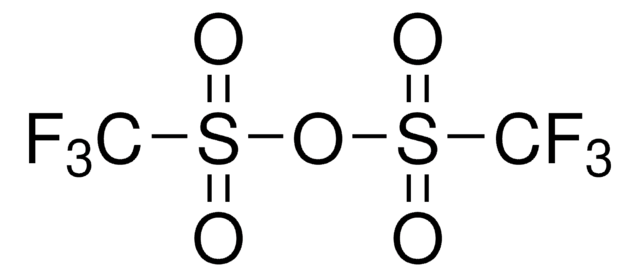
Customers Also Viewed
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service