All Photos(3)
About This Item
Empirical Formula (Hill Notation):
Cl2Pd
CAS Number:
Molecular Weight:
177.33
MDL number:
UNSPSC Code:
12352302
NACRES:
NA.22
Recommended Products
form
solid
Quality Level
composition
~ 4 wt.% loading of catalyst
reaction suitability
reagent type: catalyst
reaction type: Cross Couplings
SMILES string
Cl[Pd]Cl
InChI
1S/2ClH.Pd/h2*1H;/q;;+2/p-2
InChI key
PIBWKRNGBLPSSY-UHFFFAOYSA-L
Related Categories
General description
Palladium(II) chloride (PdCl2) reacts with unsubstituted or alkyl-substituted cyclic ketones in the CO atmosphere to afford acyclic diesters and acyclic chloro-substituted monoesters. PdCl2 reacts with N,N-dimethylallylamine in methanol to afford di-μ-chloro-bis(2-methoxy-3-N,N-dimethylaminopropyl)dipalladium(II). Palladium dichloride, acetate and acetylacetonate in the [bmim][BF4] or [bmim][PF6] (where[bmim]+ = 1-butyl-3-methylimidazolium cation) ionic liquids have been employed for the hydrodimerization of butadiene to form octa-2,7-dien-1-ol.
Application
Application Guide for Palladium Catalyzed Cross-Coupling Reactions
Used in the synthesis of semiconducting metal-containing polymers in which the polypyrrole backbone has a conformational energy minimum and is nearly planar.
Used in the synthesis of semiconducting metal-containing polymers in which the polypyrrole backbone has a conformational energy minimum and is nearly planar.
Palladium(II) chloride (PdCl2) was used in the following studies:
ChemBeads are chemical coated glass beads. ChemBeads offer improved flowability and chemical uniformity perfect for automated solid dispensing and high-throughput experimentation. The method of creating ChemBeads uses no other chemicals or surfactants allowing the user to accurately dispense sub-milligram amounts of chemical.
Learn more about ChemBeads products
For larger scale uses, product also available in powdered form (205885) & (520659)
- As catalyst for the carbonylation of organic tellurides by reaction with carbon monoxide.
- As a catalyst along with Cu(II) for the deamination of phenethylamines to phenyl substituted pyrroles.
- Together with PEG 300, promoted efficient Suzuki-coupling of aryl chlorides with aryl boronic acids.
ChemBeads are chemical coated glass beads. ChemBeads offer improved flowability and chemical uniformity perfect for automated solid dispensing and high-throughput experimentation. The method of creating ChemBeads uses no other chemicals or surfactants allowing the user to accurately dispense sub-milligram amounts of chemical.
Learn more about ChemBeads products
For larger scale uses, product also available in powdered form (205885) & (520659)
Other Notes
High-Throughput Reaction Screening with Nanomoles of Solid Reagents Coated on Glass Beads
Versatile Methods to Dispense Sub-Milligram Quantities of Solids using Chemical Coated Beads for High-Throughput Experimentation
ChemBead Enabled High-Throughput Cross-Electrophile Coupling Reveals a New Complementary Ligand
Versatile Methods to Dispense Sub-Milligram Quantities of Solids using Chemical Coated Beads for High-Throughput Experimentation
ChemBead Enabled High-Throughput Cross-Electrophile Coupling Reveals a New Complementary Ligand
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Carc. 1B Inhalation - Eye Dam. 1 - Met. Corr. 1 - Skin Sens. 1
WGK
WGK 3
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Customer Support.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Prastika Krisma Jiwanti et al.
Physical chemistry chemical physics : PCCP, 21(28), 15297-15301 (2019-04-17)
In recent years, boron-doped diamond (BDD) has been utilized as an electrode for the electrochemical reduction of CO2, and several reports have been published on this. The wide potential window of BDD enables the hydrogen evolution reaction, which competes with
Srinu Nagireddi et al.
Environmental science and pollution research international, 27(20), 24614-24626 (2019-07-25)
Considering combinatorial optimality of functional group analysis, speciation, solution chemistry complexity, Pd(II) adsorption-desorption characteristics, this article addresses the competence and efficacy of anion exchange resins namely Amberlite IRA958, Dowex Marathon MSA, Lewatit TP214, and Amberlyst A21 commercial resins. Based on
Feng-Quan Yuan et al.
Chemical communications (Cambridge, England), 47(18), 5289-5291 (2011-04-01)
We present a PdCl(2)-catalyzed protocol for highly efficient allylation and benzylation of a rich variety of N-, O-, and S-containing heteroarenes under base/acid, additive, and ligand-free conditions. The method represents the very few examples for simple, universally applicable, clean, and
Chun Liu et al.
Organic & biomolecular chemistry, 9(4), 1054-1060 (2010-12-15)
A simple and efficient protocol has been developed for the PdCl(2)-catalyzed ligand-free and aerobic Suzuki reaction of aryl bromides or nitrogen-based heteroaryl bromides with arylboronic acids in good to excellent yields in aqueous ethanol. A systematic investigation on the effect
[(RCN)2PdCl2]-catalyzed E/Z isomerization of alkenes: a non-hydride binuclear addition-elimination pathway.
Emily H P Tan et al.
Angewandte Chemie (International ed. in English), 50(41), 9602-9606 (2011-09-23)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service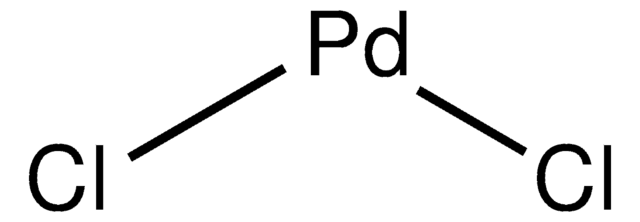

![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II) ChemBeads](/deepweb/assets/sigmaaldrich/product/structures/276/683/e8a8b162-1e17-467b-8eb6-f4180a60a1b9/640/e8a8b162-1e17-467b-8eb6-f4180a60a1b9.png)
![Dichloro[bis(2-(diphenylphosphino)phenyl)ether]palladium(II) 95%](/deepweb/assets/sigmaaldrich/product/structures/311/408/0ee427f0-19c0-413a-8f38-827359ddbcac/640/0ee427f0-19c0-413a-8f38-827359ddbcac.png)



