E1036
116-9e
≥98% (HPLC), powder
Synonym(e):
116-9e, 4-[1,1′-Biphenyl]-4-yl-3,4-dihydro-6-methyl-2-oxo-5-[(phenylmethoxy)carbonyl]-1(2H)-pyrimidinehexanoic acid
About This Item
Empfohlene Produkte
Qualitätsniveau
Assay
≥98% (HPLC)
Form
powder
Farbe
white to off-white
Löslichkeit
DMSO: >20 mg/mL
Lagertemp.
2-8°C
SMILES String
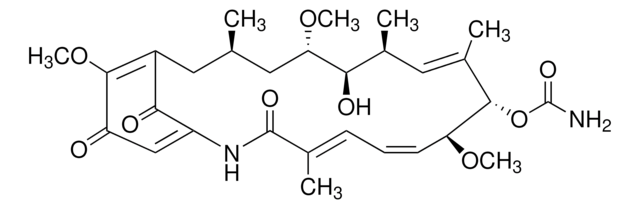
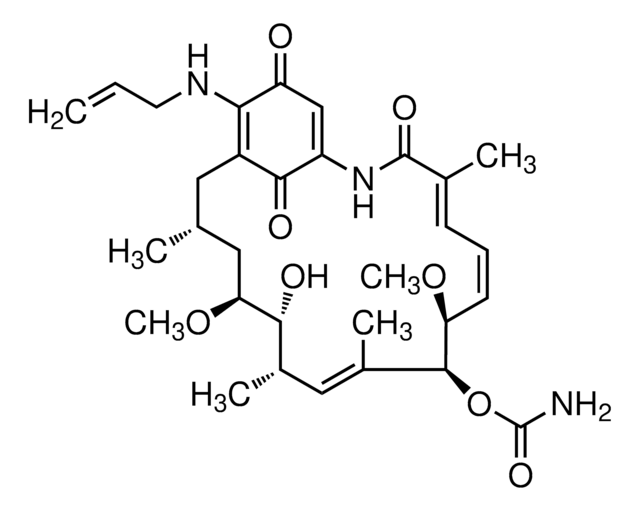
CC1=C(C(NC(=O)N1CCCCCC(O)=O)c2ccc(cc2)-c3ccccc3)C(=O)OCc4ccccc4
InChI
1S/C31H32N2O5/c1-22-28(30(36)38-21-23-11-5-2-6-12-23)29(32-31(37)33(22)20-10-4-9-15-27(34)35)26-18-16-25(17-19-26)24-13-7-3-8-14-24/h2-3,5-8,11-14,16-19,29H,4,9-10,15,20-21H2,1H3,(H,32,37)(H,34,35)
InChIKey
GHFQWLNXJMUCGC-UHFFFAOYSA-N
Anwendung
Biochem./physiol. Wirkung
The same compound by a different name, MAL2-11B, has been found to inhibit the activity of a viral J-domain protein, large tumor antigen (TAg). MAL2-11B inhibited both TAg′s endogenous ATPase activity and the TAg-mediated activation of Hsp70.
Signalwort
Danger
H-Sätze
P-Sätze
Gefahreneinstufungen
Acute Tox. 3 Oral - Aquatic Acute 1 - Aquatic Chronic 1
Lagerklassenschlüssel
6.1C - Combustible, acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Analysenzertifikate (COA)
Suchen Sie nach Analysenzertifikate (COA), indem Sie die Lot-/Chargennummer des Produkts eingeben. Lot- und Chargennummern sind auf dem Produktetikett hinter den Wörtern ‘Lot’ oder ‘Batch’ (Lot oder Charge) zu finden.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.
![PERK-Inhibitor I, GSK2606414 GSK2606414 is a cell-permeable, highly potent inhibitor of EIF2AK3/PERK (IC₅₀ = 0.4 nM; [ATP] = 5 µM). Targets PERK in its inactive DFG conformation at the ATP-binding region.](/deepweb/assets/sigmaaldrich/product/structures/180/559/efa716dc-d5fe-4339-a6f0-0103084fc04a/640/efa716dc-d5fe-4339-a6f0-0103084fc04a.png)