03960590
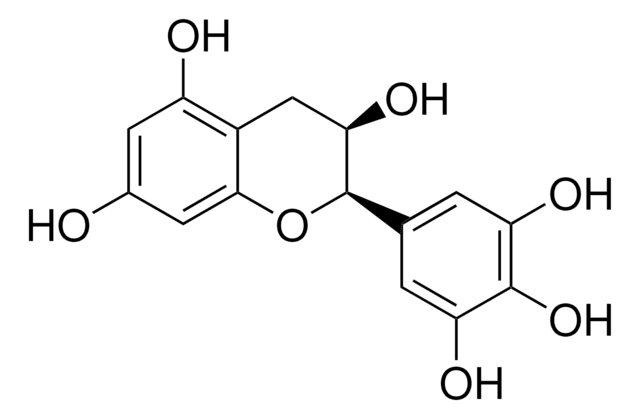
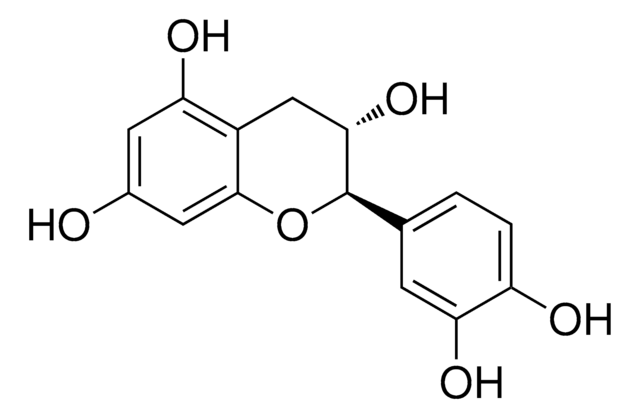
Epigallocatechin
primary reference standard
Synonym(e):
(−)-Epigallocatechin, (−)-cis-2-(3,4,5-Trihydroxyphenyl)-3,4-dihydro-1(2H)-benzopyran-3,5,7-triol, (−)-cis-3,3′,4′,5,5′,7-Hexahydroxy-flavan
About This Item
Empfohlene Produkte
Qualität
primary reference standard
Haltbarkeit
limited shelf life, expiry date on the label
Hersteller/Markenname
HWI
Anwendung(en)
food and beverages
Lagertemp.
−20°C
SMILES String
O[C@@H]1Cc2c(O)cc(O)cc2O[C@@H]1c3cc(O)c(O)c(O)c3
InChI
1S/C15H14O7/c16-7-3-9(17)8-5-12(20)15(22-13(8)4-7)6-1-10(18)14(21)11(19)2-6/h1-4,12,15-21H,5H2/t12-,15-/m1/s1
InChIKey
XMOCLSLCDHWDHP-IUODEOHRSA-N
Suchen Sie nach ähnlichen Produkten? Aufrufen Leitfaden zum Produktvergleich
Verwandte Kategorien
Allgemeine Beschreibung
Exact content by quantitative NMR can be found on the certificate.
Anwendung
Biochem./physiol. Wirkung
WGK
WGK 3
Flammpunkt (°F)
Not applicable
Flammpunkt (°C)
Not applicable
Choose from one of the most recent versions:
Analysenzertifikate (COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Besitzen Sie dieses Produkt bereits?
In der Dokumentenbibliothek finden Sie die Dokumentation zu den Produkten, die Sie kürzlich erworben haben.
Kunden haben sich ebenfalls angesehen
Unser Team von Wissenschaftlern verfügt über Erfahrung in allen Forschungsbereichen einschließlich Life Science, Materialwissenschaften, chemischer Synthese, Chromatographie, Analytik und vielen mehr..
Setzen Sie sich mit dem technischen Dienst in Verbindung.